Sampling & Collection
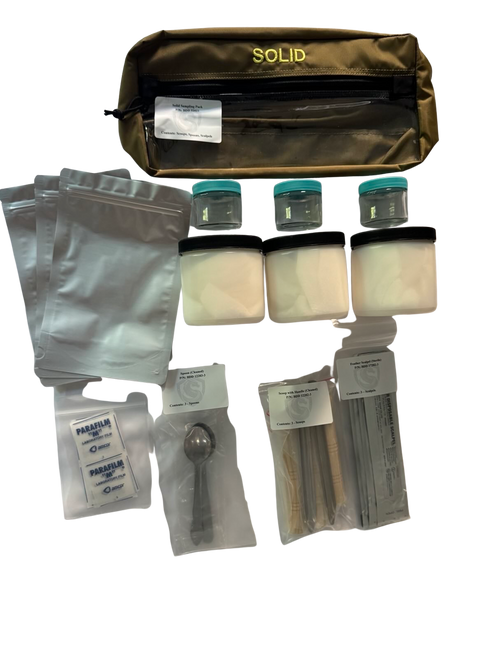
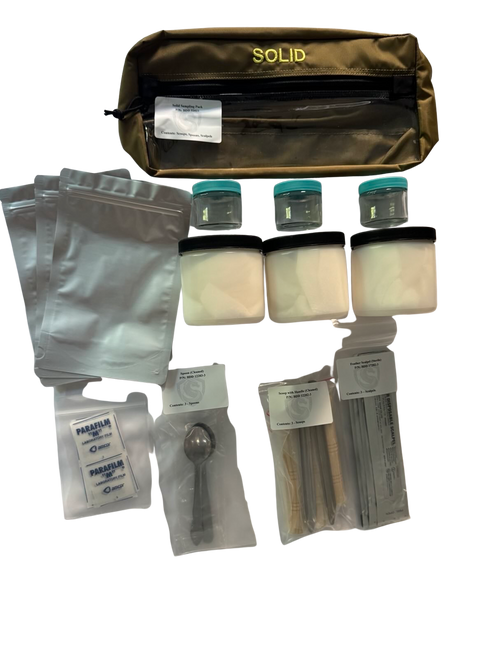
CBRN Sampling Kits and Collection Overview
CBRN sampling is the controlled collection of environmental or material samples during chemical, biological, radiological, or nuclear incidents so qualified laboratories can identify hazards and support response, public safety, investigative, and recovery decisions. Reliable sampling data is necessary to confirm contamination, assess risk, and determine appropriate protective actions, decontamination requirements, or remediation strategies.
The U.S. Environmental Protection Agency identifies CBRN sampling as a critical component of emergency response and consequence management, supporting environmental protection and public health decision-making during hazardous incidents.
Source: U.S. Environmental Protection Agency – CBRN Emergency Response Resources
https://www.epa.gov/emergency-response/chemical-biological-radiological-and-nuclear-quick-reference-guides
CBRN sampling may involve multiple environmental matrices, including air, liquids, solids such as soil or powders, and surface residues. Sampling plans define the target matrix, collection method, contamination controls, documentation requirements, and chain-of-custody procedures to preserve analytical integrity. EPA sampling guidance emphasizes defining objectives and quality controls to prevent cross-contamination and preserve sample validity.
Source: U.S. EPA SW-846 Sampling Plans Guidance
https://www.epa.gov/hw-sw846/chapter-nine-sw-846-compendium-sampling-plans
Certified Cleaning and Biological Sterility
Preventing external contamination is essential for credible CBRN sampling. Sampling equipment that is not properly cleaned or sterile can introduce interference that compromises analytical results or leads to false conclusions.
Blue Dragon Defense certifies that its CBRN sampling kits are chemically cleaned and biologically sterile prior to packaging to reduce the risk of outside interference during sample collection.
Chemical cleaning refers to the removal of residual chemical contaminants from sampling components so that collected samples reflect conditions at the incident site rather than artifacts from manufacturing or handling. The EPA emphasizes equipment cleanliness and contamination control as part of environmental sampling quality assurance practices.
Source: U.S. EPA – Sample Handling and Custody Guidance
https://www3.epa.gov/ttnamti1/files/ambient/pm25/qa/vol2sec08.pdf
Biological sterility refers to the absence of viable microorganisms that could contaminate biological samples or interfere with laboratory analysis. The Centers for Disease Control and Prevention defines sterilization as a process that eliminates all forms of microbial life and identifies it as a core practice for preventing cross-contamination in biological sampling and laboratory contexts.
Source: U.S. Centers for Disease Control and Prevention – Sterilization and Disinfection Guidelines
https://www.cdc.gov/infectioncontrol/guidelines/disinfection/
Using chemically cleaned and biologically sterile sampling kits supports data integrity when results are used for public safety decisions, regulatory evaluations, or forensic and investigative purposes.
Standards and Interoperability Context
CBRN equipment and procedures must function reliably across agencies and jurisdictions. The U.S. Department of Homeland Security publishes a national strategy for chemical, biological, radiological, nuclear, and explosives standards to promote interoperability, reliability, and confidence in response equipment and capabilities.
Source: U.S. Department of Homeland Security – National Strategy for CBRNE Standards
https://www.dhs.gov/national-strategy-chemical-biological-radiological-nuclear-and-explosives-cbrne-standards
Chain of Custody and Safe Sample Handling
Chain of custody is the documented record of sample control and transfer from collection through shipping and laboratory analysis. It supports traceability and defensibility when results inform regulatory, investigative, or high-consequence operational decisions.
EPA guidance outlines custody procedures, labeling, sealing, storage, and transport requirements to ensure samples remain intact and associated with correct documentation.
Source: U.S. EPA – CLP Guidance for Field Samplers
https://www.epa.gov/sites/default/files/2021-03/documents/samplers_guide_clp_guidance_for_field_samplers_november_2020.pdf